A conversation with Harbour BioMed, I-Meb Biopharma, JW Therapeutics and Legend Biotech
Founded in 2016, IPO in 2020: the fast development of these four companies is snapshot of Chinese
It is both co-incident and destiny that the four Chinese biotech companies were founded in the same year and were public 4 years later.
| Founding Year | IPO Market | IPO Date | |
Harbour BioMed | 2016 | HKEX | 2020.12 |
| I-Mab Biopharma | 2016 | NASDAQ | 2020.1 |
JW Therapeutics | 2016 | HKEX | 2020.11 |
Legend Biotech | 2016 | NASDAQ | 2020.6 |
Through their pipelines, international collaborations and company vision, we could clearly see the rising of these Chinese biotech superstars with global ambitions.
Pipelines
All four companies have build strong pipelines with late stage clinical products
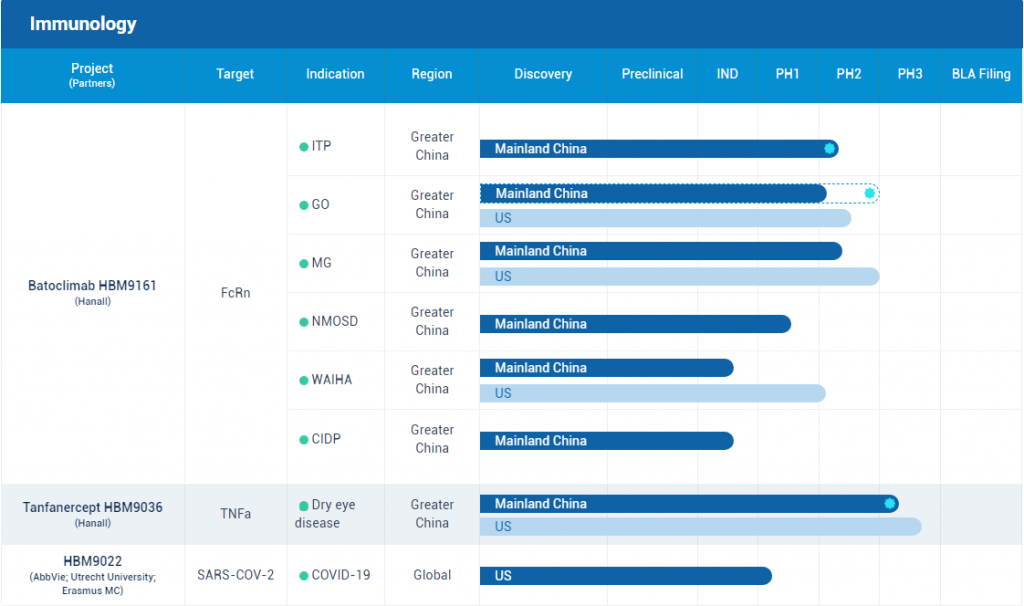
Pipeline of Harbour BioMed – Immunology (Source: official Website)

Pipeline of Harbour BioMed – Oncology (Source: official Website, 2021.1)
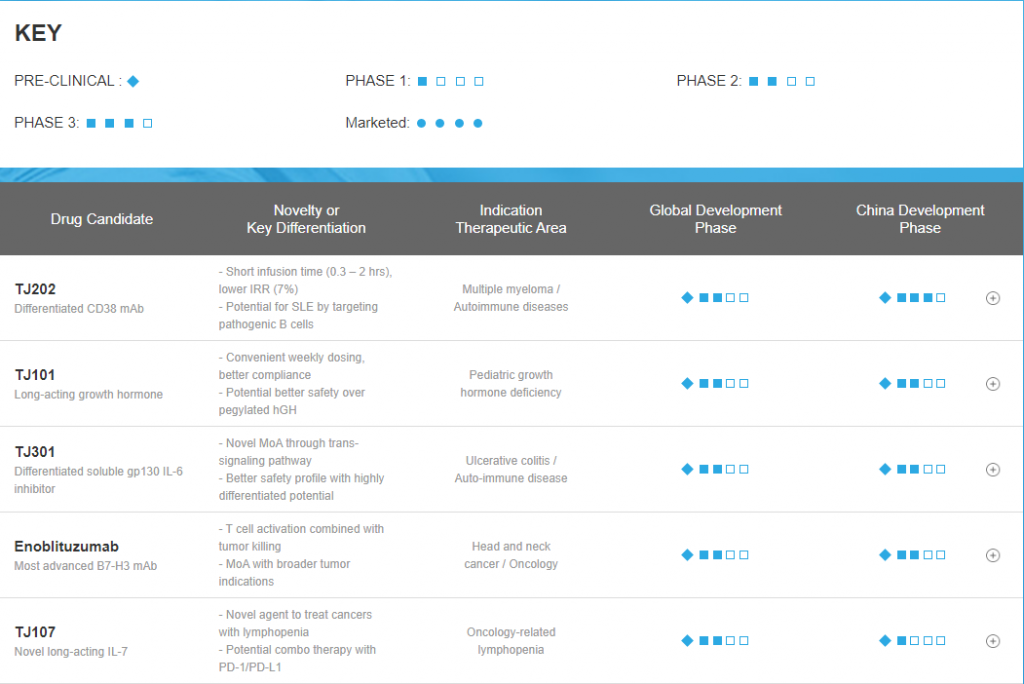
Pipeline of I-Mab Biopharma – China Market (Source: official Website, 2021.1)
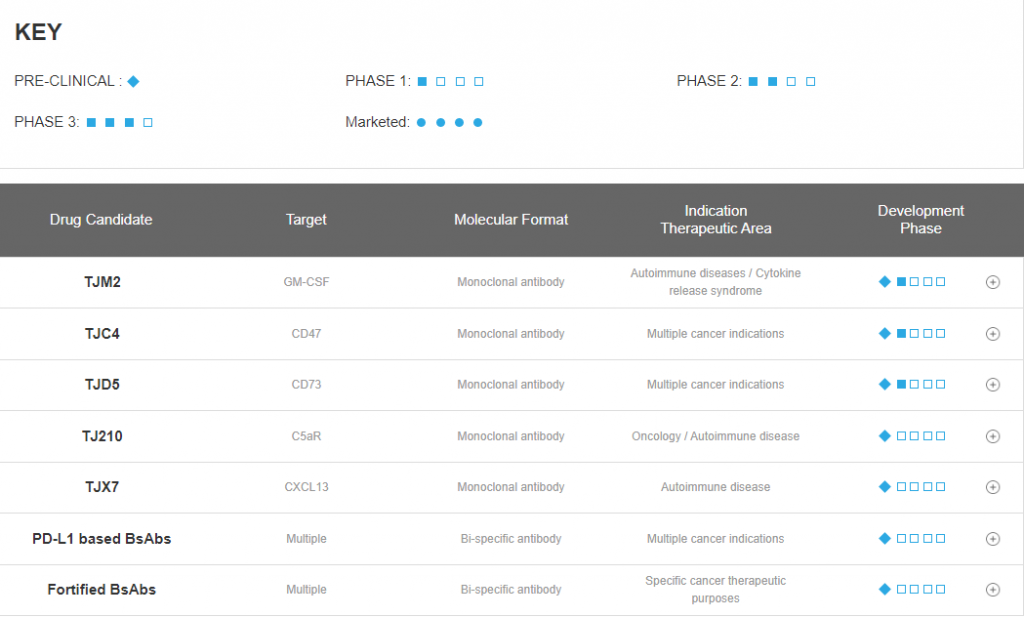
Pipeline of I-Mab Biopharma – Global Market (Source: official Website, 2021.1)
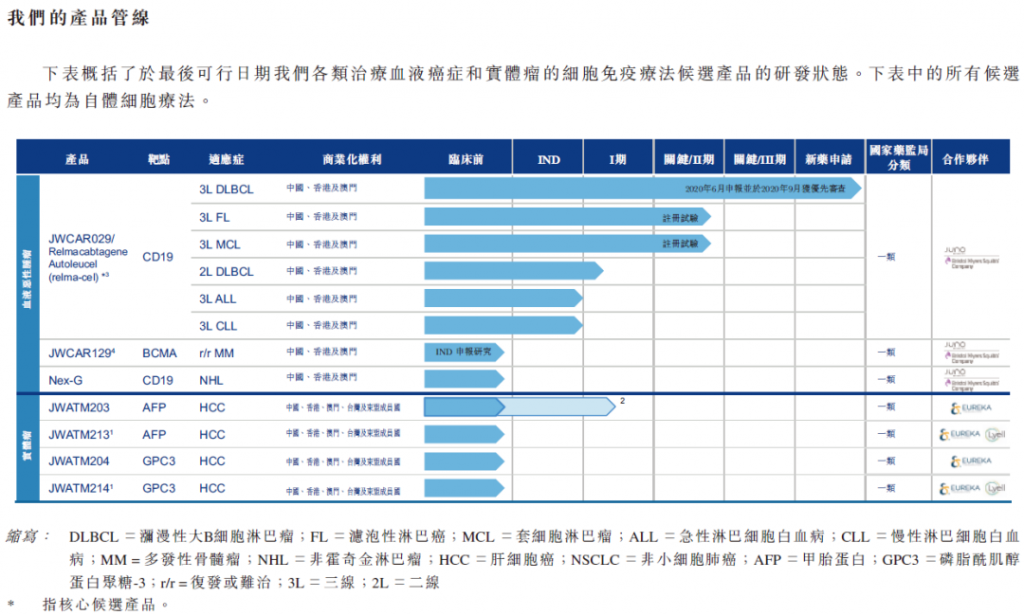
- Pipeline of JW Therapeutics (Source: JW’s IPO document, 2020.10)

Pipeline of Legend Biotech (Source: official Website, 2021.1)
Global collaborations
Harbour BioMed: multiple global partners
With H2L2, HCAb, HBICE, Single B Cell Technology platforms, Harbour has built multiple strategic partnership with domestic and international players. The Latter includes AbbVie, Utrecht University, Erasmus MC, Hanall Biopharma and Eli Lilly.
I-Mab and AbbVie
On the evening of September 4, AbbVie, a global TOP biopharmaceutical company, announced that it has reached a global strategic cooperation on the development and commercialization of the anti-cancer drug CD47 monoclonal antibody lemzoparlimab (TJC4) under development by I-Mab Biopharma. AbbVie will pay up to US$1.94 billion in down payment and milestone payments, plus no less than US$1 billion in potential bispecific antibody cooperation, with a maximum potential value of US$3 billion. This transaction not only refreshed the historical maximum amount of overseas authorized transactions by Chinese biopharmaceutical companies, but also the largest amount of authorized transactions in the global biotechnology field in the past five years.
JW Therapeutics: started as JV between Wuxi and Juno
It was founded in 2016 as a venture between Juno and Wuxi, the aim being to license cell therapy candidates from Juno’s pipeline for local development, backed by Wuxi’s local knowledge and alliances. That deal remains in place until 2026, and under it JW can continue licensing in Juno’s cell therapy projects even though ownership of that business has now moved via Celgene to Bristol Myers Squibb.
Legend Biotech and Janssen
Legend Biotech has entered into a worldwide collaboration and license agreement with Janssen Biotech, Inc., one of the Janssen Pharmaceutical Companies of Johnson & Johnson, to develop, manufacture, and commercialize LCAR-B38M (JNJ-68284528/JNJ-4528). Under the terms of the agreement, Legend and Janssen are co-developing and will co-commercialize LCAR-B38M/JNJ-4528 in select countries. Manufacturing of LCAR-B38M/JNJ-4528 for commercial distribution worldwide will be done in partnership with Janssen.
Company Overviews
Harbour BioMed
Harbour BioMed is a global, clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel antibody therapeutics in the areas of oncology and immunological diseases to address unmet patient’s needs.
We are building a robust pipeline with our integrated Harbour antibody platform, that enable us to develop highly differentiated antibodies against various disease targets with great potency and safety profiles. Our proprietary Harbour Mice® generate fully human monoclonal antibodies in the classical two heavy and two light chain (H2L2) format, as well as heavy chain only (HCAb) format. Building upon our HCAb antibodies, the HCAb-based immune cell engagers (HBICETM) are capable of delivering tumor killing effects unachievable by combination therapies. Integrated with our single B cell cloning platform, our antibody discovery engine is highly unique and efficient for development of next generation therapeutic antibodies.
I-Mab Biopharma
We believe we are uniquely positioned as a China-based global player, with a presence in Shanghai, Beijing and the US, to tap into these vast commercial opportunities. Since its founding in 2016, I-Mab has rapidly become one of the top innovative biotech companies in China, and we have successfully raised more than US$400 million in equity financing from our dedicated group of investors, including leading healthcare-focused funds. Our research and development capabilities encompass discovery, translational medicine, chemistry, manufacturing and controls (CMC) for biologics, pre-clinical development and clinical development, as well as business development functions. These integrated capabilities are strengthened by our deep understanding of China’s biologics regulatory framework combined with our direct access to extensive preclinical and clinical trial resources in China. I-Mab is now at a critical juncture to transition from a clinical-stage biotech company into a fully integrated end-to-end global biopharmaceutical company in the next few years.
We are a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel or highly differentiated biologics to treat diseases with significant unmet medical needs, particularly cancers and autoimmune disorders. Our mission is to bring transformational medicines to patients through innovation.
We see vast commercial opportunities for immuno-oncology and autoimmune biologics therapies in China and rest of the world. We were founded to capture the opportunities presented by the confluence of two major developments: the emergence of an attractive and growing biologics market in China, and revolutionary scientific breakthroughs in cancer and autoimmune disease medicines.
To achieve our mission and capitalize on these commercial opportunities, we have developed a business model built on two pillars. The first is a fast-to-market China approach, and the second a fast-to-PoC (proof of concept) global approach. Reflecting these two approaches, we have established a China Portfolio and a Global Portfolio of novel or highly differentiated clinical and preclinical assets in the most exciting therapeutic areas of immuno-oncology and immuno-inflammation. Through our internal research and development efforts, and in-licensing arrangements with global pharmaceutical and biotech companies, we have built an innovative pipeline of more than ten assets in clinical and preclinical stages of development. We are currently running three Phase 1 clinical trials in the US and China, and a further four trials, mostly Phase 2 or 3, in China.
JW Therapeutics
JW (Cayman) Therapeutics Co. Ltd (the “Company”) is a global leading clinical stage cell therapy platform company. The Company’s vision is to develop best-in-class and/or first-in-class cell therapies for the China market to transform the treatment of cancer for Chinese patients. Since the establishment of JW Shanghai in 2016 , it has built an integrated platform focused on developing, manufacturing and commercializing breakthrough cell-based immunotherapies for hematological cancers and solid tumors.
The company is a pioneer in China for the development of cell-based immunotherapy, a field which represents a paradigm shift and the latest advancement in the treatment of cancer. Cell-based immunotherapies, including CAR-T treatments, are an innovative treatment method that uses human immune cells to fight cancer. Supported by multiple clinical studies, cell-based immunotherapies could lead to long-lasting remissions of B-cell lymphomas and leukemias which are refractory to other treatments.
Relmacabtagene autoleucel (“relma-cel”), its lead product candidate, is an anti-CD19 CAR-T therapy for relapsed or refractory (“r/r”) B-cell lymphoma, and in June 2020 the National Medical Products Administration (“NMPA”) accepted for review its NDA relating to relma-cel as a third-line treatment for DLBCL. Relma-cel is expected to be the first CAR-T therapy to be approved as a Category 1 biologics product in China, and has potential to be a best-in-class CAR-T therapy.
Legend Biotech
Legend Biotech is a global clinical-stage biopharmaceutical company engaged in the discovery and development of novel cell therapies for oncology and other indications. Our team of over 800 employees across the United States, China and Europe, along with our differentiated technology, global development, and manufacturing strategies and expertise, provide us with the strong potential to discover, develop, and manufacture cutting -edge cell therapies for patients in need. We are engaged in a strategic collaboration to develop and commercialize our lead product candidate, ciltacabtagene autoleucel, an investigational BCMA targeted CAR-T cell therapy for patients with multiple myeloma. This candidate is currently being studied in registrational clinical trials.
A conversation with the four Chinese Biotech Superstars
However, IPO is just another start for these companies rather than destination. With no current product on the market, all of them are facing challenges and great opportunities in the dawn of Chinese pharmaceutical innovation market.
We’ve invited leaders from these four companies to share their vision in building the company and discuss questions including:
- Pricing is under pressure in China, how do they build portfolio?
- What is their business model? How do they generate value for investors in the long run? How do they position themselves, a China Biotech or global biotech with China angle?
- Oncology has been the hot market in China, and it all plays an important role in their current endeavor. What is next?
- What are their predictions for 2021? How do they view the influence of COVID-19 on the Biotech industry?
Let’s hear how leaders from these companies would say.
Registration Link: https://us02web.zoom.us/webinar/register/WN_bkP1S82oREyl-v_hDX0Zsg

Source:
- Official Websites
- NASDAQ/HKEX company profiles
- JW’s IPO document