Attention deficit hyperactivity disorder (ADHD) is one of the most common mental disorders affecting children. Guidelines within the American Psychiatric Association’s Diagnostic and Statistical Manual, Fifth edition (DSM-5) are used to diagnose ADHD. According to the DSM criteria, as cited by the CDC, people with ADHD show a persistent pattern of inattention and/or hyperactivity–impulsivity that interferes with functioning or development:
1. Inattention: Six or more symptoms of inattention for children up to age 16 years, or five or more for adolescents age 17 years and older and adults; symptoms of inattention have been present for at least 6 months, and they are inappropriate for developmental level:
• Often fails to give close attention to details or makes careless mistakes in schoolwork, at work, or with other activities.
• Often has trouble holding attention on tasks or play activities.
• Often does not seem to listen when spoken to directly.
• Often does not follow through on instructions and fails to finish schoolwork, chores, or duties in the workplace (e.g., loses focus, side-tracked).
• Often has trouble organizing tasks and activities.
• Often avoids, dislikes, or is reluctant to do tasks that require mental effort over a long period of time (such as schoolwork or homework).
• Often loses things necessary for tasks and activities (e.g. school materials, pencils, books, tools, wallets, keys, paperwork, eyeglasses, mobile telephones).
• Is often easily distracted.
• Is often forgetful in daily activities.
2. Hyperactivity and Impulsivity: Six or more symptoms of hyperactivity-impulsivity for children up to age 16 years, or five or more for adolescents age 17 years and older and adults; symptoms of hyperactivity-impulsivity have been present for at least 6 months to an extent that is disruptive and inappropriate for the person’s developmental level:
• Often fidgets with or taps hands or feet, or squirms in seat.
• Often leaves seat in situations when remaining seated is expected.
• Often runs about or climbs in situations where it is not appropriate (adolescents or adults may be limited to feeling restless).
• Often unable to play or take part in leisure activities quietly.
• Is often “on the go” acting as if “driven by a motor”.
• Often talks excessively.
• Often blurts out an answer before a question has been completed.
• Often has trouble waiting their turn.
• Often interrupts or intrudes on others (e.g., butts into conversations or games)
In China, the total prevalence of ADHD in school-age children is estimated at 3%-10%. The global incidence of ADHD is 1.7-16%, with the incidence of school-age children at 3%-5%. The adult incidence rate is 4% to 5%. About 50% to 70% of children with ADHD will continue to have the disorder into adulthood.
The annual direct and indirect costs of ADHD in the United States amount to US$21.87 billion. The global ADHD market ranks ninth among central nervous system diseases, with an annual growth rate of 8% every year. In China, children account for about 17% of the total population. Due to the disorder’s high incidence, the patient population is huge.
A new drug with an early onset, long duration time
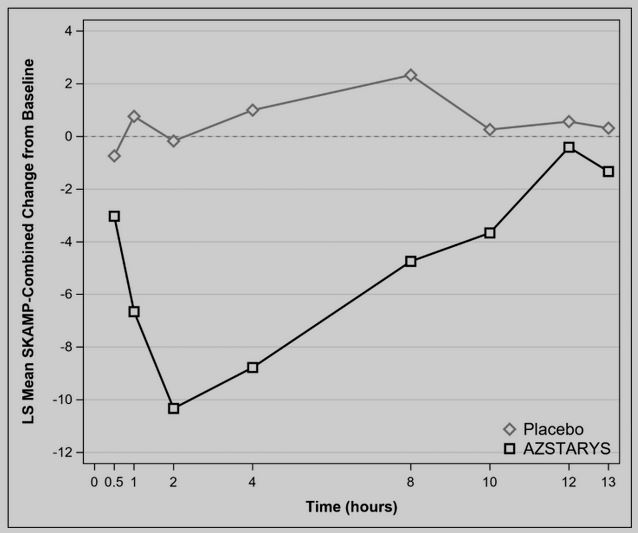
Recently, KemPharm and Corium jointly announced that the FDA has approved the once-a-day oral capsule Azstarys for the treatment of children with ADHD over 6 years old. This is the first ADHD therapy that contains a d-methylphenidate “prodrug”, which can maintain the effect for a long time while having a rapid onset. It is expected that the drug will be commercialized in the United States as early as the second half of 2021.
Azstarys is a central nervous system agonist, which contains 30% rapid-release d-methylphenidate (d-MPH) hydrochloride, 70% serdexmethylphenidate (SDX), and a prodrug of d-MPH.
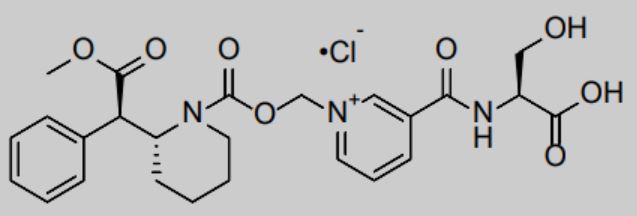
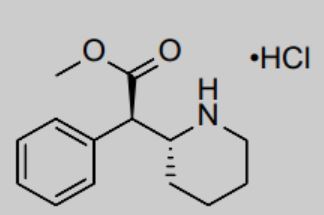
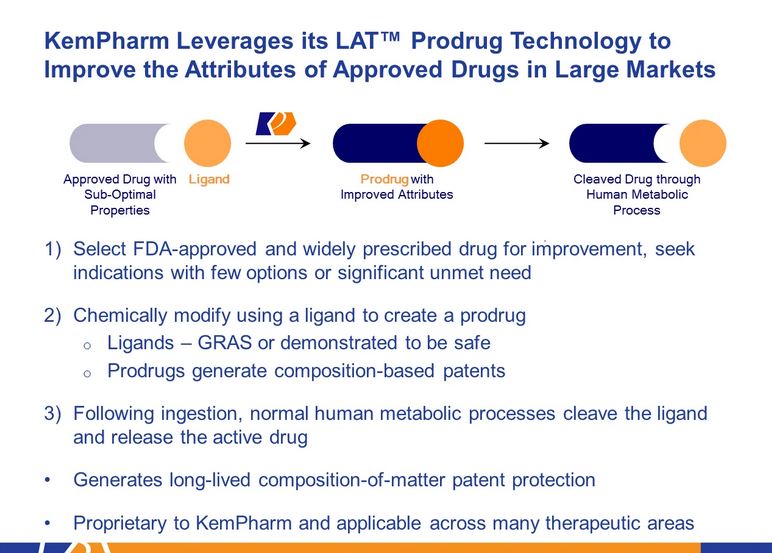
KemPharm uses its proprietary ligand activated therapy (LAT®) technology to link d-MPH with another compound to create a prodrug. This drug is initially inactive after taking it, but after being cut by metabolic enzymes in the gastrointestinal tract, it slowly releases the active ingredients, thereby maintaining the efficacy for a long time. This prolonged release can also prevent drugs from being abused.
Azstarys’ special dual role solves the market’s unmet needs for early onset ADHD medication with long-term treatment.
The new molecules created by LAT® technology have better safety and effectiveness than comparable drugs.
“This innovative therapy with a dual mechanism of action uses d-methylphenidate in combination with its prodrug for the first time. It is good news for the families and doctors of patients who want to choose the right ADHD therapy,” said lead clinical trial researcher, Dr. Ann Childress, President of Xue Medical Center.
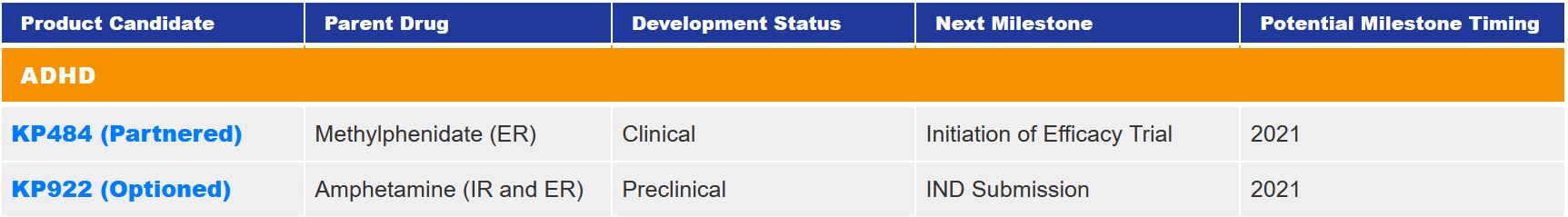
KemPharm’s ADHD drug pipeline
KP484, also composed of serdexmethylphenidate (SDX), is linked with d-methylphenidate (d-MPH) to form a prodrug. The preclinical and clinical studies of KP484 show that compared with the most commonly used ADHD treatment drug methylphenidate, KP484 can produce a longer d-MPH release time. Based on this release characteristic, it is believed that KP484 will have a large and growing market space for adult ADHD patients.
KP922 is currently in the pre-clinical research stage and is expected to enter clinical trials in 2021.
Common treatment drugs for ADHD
The FDA has approved more than 30 kinds of drugs to treat ADHD. Commonly used treatment drugs in China include central stimulants, such as methylphenidate hydrochloride, and non-stimulant drugs, such as atomoxetine hydrochloride.
Methylphenidate

The date when methylphenidate was first approved by the FDA was December 5, 1955, under the trade name Ritalin, with a dosage form as a regular-release tablet. After the 1970s, as the sustained and controlled release technology became more mature, Novartis developed a sustained-release prescription, which was approved by the FDA in 1982 under the trade name Ritalin SR. Ritalin SR is a sustained-release tablet prepared with zein combined with stearyl alcohol, mineral oil, povidone and other materials. Compared with normal-release tablets, Ritalin SR lasts longer, up to 8 hours. However, it is still not possible to administer once a day, and it can only be swallowed whole, which is not particularly convenient for children. Fortunately, the diameter of Ritalin tablets is only 7-8mm, and older children can still take it. Novartis has sold for more than half a century.
Atomoxetine
Atomexetine is selective norepinephrine (noradrenaline) reuptake inhibitor that is not classified as a stimulant, with a long acting time and good tolerance. The mechanism of action for the treatment of ADHD is that it can increase the level of norepinephrine, increase normal phase transmission, and increase the level of dopamine in the prefrontal cortex without increasing the dopamine activity in the striatum, so it will not induce symptoms of tics or increased movement disorders. It is sold under the brand name Strattera, among others.
Amphetamine
Guanfacine
Guanfacine was marketed in the United States in September 2009 and in the European Union in September 2015 for the treatment of ADHD. The approval of guanfacine hydrochloride sustained-release tablets marks a major development. Guanfacine hydrochloride can directly bind to receptors in the prefrontal cortex, and the excitement of the postsynaptic α2a -receptor may enhance working memory, reduce distraction, improve attention regulation, increase behavioral inhibition, and enhance impulsivity control, as well as having other effects.
Guanfacine hydrochloride is one of the top 5 ADHD drugs.
Clonidine
In October 2010, the U.S. FDA approved the Clonidine Hydrochloride Sustained-Release Tablet Kapvay developed by Shiono Yoshihide Pharmaceutical Co., Ltd. for single-use or auxiliary excitatory drugs to treat ADHD in children from 6 to 17 years old to adolescents. In China, clonidine hydrochloride tablets are antihypertensive drugs that can be used to treat high blood pressure and to help opioid addiction symptoms.
Summary
Azstarys is the first ADHD therapy containing a d-methylphenidate “prodrug”. At present, there are many kinds of drugs for the treatment of ADHD, the efficacy is highly dependent on the drug. Furthermore, some of the drugs include a risk for abuse. Therefore, the research and development of drugs for the treatment of ADHD has long been a hot topic in the medical field. The approval of Azstarys is promising, and it is expected to change the treatment landscape of ADHD patients.