Fast track for medical device registration and approval
In recent years, the NMPA has issued documents to provide direction and guidance for the reform of medical device review and approval.
The National Medical Products Administration issued initial documentation in March 2014 and revised it in November 2018. The new procedure sets up a fast-track review and approval pathway for innovative medical devices. It also requires the establishment of an examination office to carry out the examination of certain applications for innovative medical devices.
The Medical Inspection Center of the China Food and Drug Administration oversees the procedures for the special review of innovative medical devices. It has formulated the “Innovative Medical Device Special Review Application Review Operation Specification” to further improve work processes, refine review requirements, and optimize the review method. The Medical Inspection Center also standardized the requirements for the preparation and submission of application materials for innovative applications and provided specific guidance for relevant both product applicants and R&D personnel. Meanwhile, an online appointment consultation platform and a smooth and standardized communication channel with applicants was established.
These improvements to approval procedures should accelerate the speed of medical device approval. After entering this channel, relevant agencies mainly apply early intervention, dedicated personnel, and full guidance. They prioritize reviewing the application under the condition that approval standards and procedures are maintained.
Taking the application of Chinese medical devices as an example, the provincial drug supervision bureaus complete the preliminary review within 20 working days. The NMPA must then issue a review opinion within 40 working days. The application to registration should be completed in about 15 weeks. Products registered this way take an average of 83 days less time to register than previously.
Innovative medical devices continue to emerge
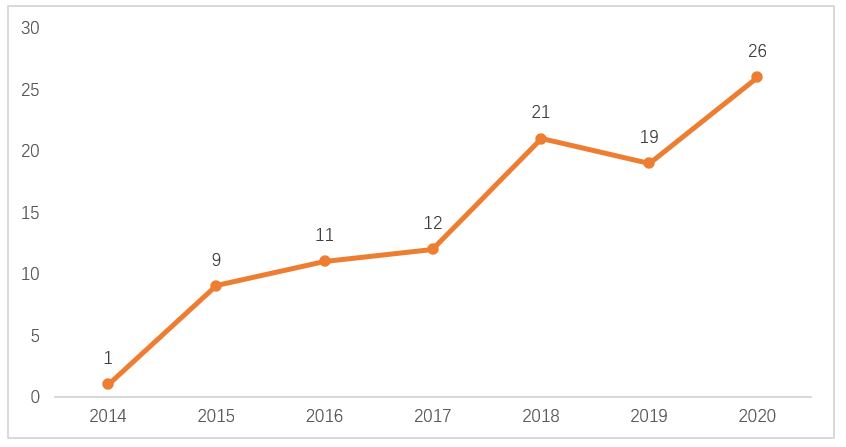
Since the opening of the fast-track approval process for innovative medical devices in 2014, as of the end of December 2020, 1,471 products applied and 292 products were included as innovative medical devices, accounting for about 20% of all applications. In recent years, the number of innovative medical devices approved has increased almost year by year, with 1 innovative device approved in 2014, 9 approved in 2015, 11 approved in 2016, 12 approved in 2017, 21 approved in 2018, 19 approved in 2019, and 26 approved in 2020.
Source: NMPA
Innovative medical devices cover multiple fields
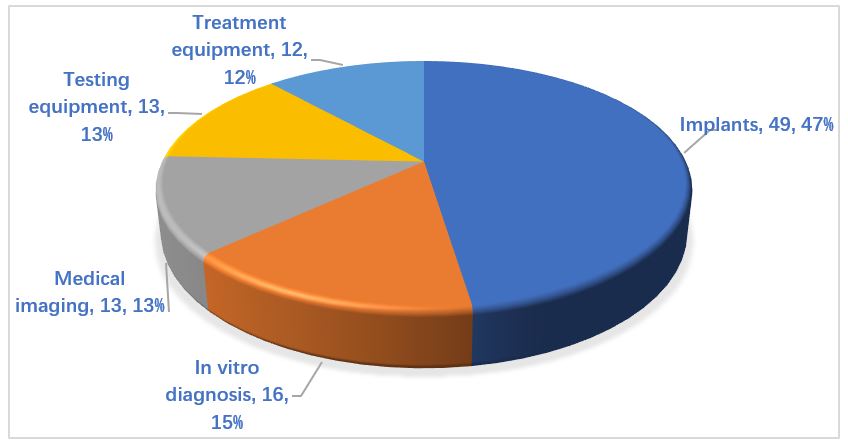
In terms of product types, the majority of innovative medical devices in China are implants, accounting for about 50% of the total. Meanwhile, the number of testing, treatment, medical imaging, and in vitro diagnostic products is roughly equivalent, between 12% and 15% of the total each.
Source: NMPA
Implants
The implants are mainly within the cardiovascular, CNS, and optometry fields.
Cardiovascular implants include pacemakers, cardiac stents, artificial heart valves, left atrial appendage closure systems, drug-eluting stents, and balloons.
The percutaneous interventional artificial heart valve system developed by Venus Medtech in Hangzhou is the first self-expanding percutaneous interventional artificial heart valve system in China. It is suitable for symptomatic, calcified, and severe aortic valve stenosis, and is not suitable patients undergoing routine surgery to replace their valves. The branched aortic stent graft and delivery system developed by MicroPort in Shanghai is the first domestically approved branched aortic stent graft for the treatment of patients with thoracic aortic dissection. The bioabsorbable coronary rapamycin-eluting stent system developed by Bio Huaan in Shandong is used to treat intravascular stenosis in patients with primary coronary atherosclerosis, improve coronary blood flow and prevent restenosis. occur. Compared with traditional metal drug stents, the final degradation products of this product are water and carbon dioxide, reducing the trouble of late thrombosis.
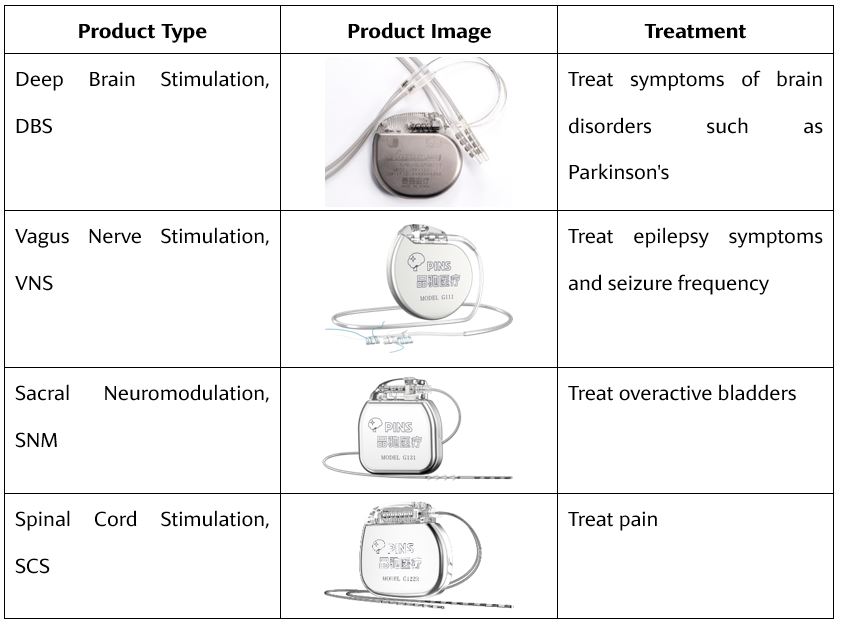
CNS products can be subdivided into deep brain electrical stimulation, vagus nerve stimulation, sacral nerve electrical stimulation, and spinal cord electrical stimulation. The dual-channel implantable deep brain electrical stimulation pulse generator kit, implantable deep brain electrical stimulation electrode lead set and implanted deep brain electrical stimulation extension lead set developed by Sceneray in Suzhou are used for treatment of patients with advanced primary Parkinson’s who cannot effectively control the symptoms of dyskinesias. The implantable vagus nerve stimulation pulse device and the implantable vagus nerve stimulation electrode device developed by Pins Medical in Beijing are the first domestic products that use vagus nerve stimulation to treat epilepsy.
Medical imaging
Medical imaging devices mainly include positron emission and X-ray computed tomography scanning systems, imaging catheters, and CT image-assisted detection software.
The positron emission and X-ray computed tomography imaging device developed by MinFound combines X-ray computed tomography (CT) and positron emission computed tomography (PET). The relevant information generated by the scan can be used for brain cancer detection, diagnosis, treatment and efficacy evaluation as well as for cardiovascular diseases. The positron emission tomography and magnetic resonance imaging system developed by United Imaging in Shanghai includes two parts: positron emission tomography and magnetic resonance. The high resolution, high sensitivity, and low radiation dosage have significant advantages in the diagnosis of head and neck cancer, abdominal cancer, degenerative neuropathy, systemic lymphoma, and other diseases.
Testing equipment
Testing equipment mainly includes blood flow reserve fraction measurement, nucleic acid testing, cardiac electrophysiological mapping, automatic chemiluminescence immunoassay analyzer, blood glucose meter, and auxiliary testing software.
The three-dimensional cardiac electrophysiological mapping system developed by MicroPort EP in Shanghai combines an intracardiac electrogram with spatial structure to help the surgeon better complete the ablation treatment of complex arrhythmias. The coronary angiography blood flow reserve measurement system developed by RainMed in Suzhou has created a new blood flow reserve calculation method and detection method which can effectively improve the accuracy of diagnosis, reduce the risk of surgery, help promote the popularization of FFR indicators to diagnose myocardial ischemia, and assist doctors in formulating precise treatment strategies suitable for their own conditions. The nucleic acid amplification and detection analyzer developed by USTAR in Hangzhou is based on the three-section magnetic guide extraction technology and the principle of fluorescent polymerase chain reactions. It is used together with the matching detection device for qualitative detection of mycobacterium tuberculosis complex nucleic acid in human samples.
Treatment equipment
Treatment equipment mainly includes medical accelerators, surgical navigation and positioning systems, cryoablation surgical systems, plasma surgical treatments, and cancer radiotherapy.
The medical electronic linear accelerator developed by Space in Guangdong can perform fixed-beam or moving-beam image-guided intensity-modulated radiotherapy for solid tumors, as well as X-ray simulation positioning and verification of treatment plans. The neurosurgery navigation and positioning system developed by Sinovation in Beijing is used for the positioning and orientation of neurosurgery stereotactic surgery. The puncture surgery navigation device developed by EDDA Technology in Suzhou is the first domestic puncture surgery navigation device for adult lung and abdominal soft tissue solid organs. It is used for adult lung and abdominal solid organ puncture operations. The tumor electric field treatment instrument developed by Novokuler is the first medical device in China that uses the principle of electric field to inhibit cell division. It can be used as a treatment method for patients with glioblastoma after surgery, radiotherapy, or chemotherapy.
In Vitro Diagnostics
In vitro diagnostics include single-gene detection and multi-gene detection. For example, there is the Septin9 gene methylation detection kit (PCR fluorescent probe method) developed by BioChain in Beijing and the EGFR gene mutation detection kit (multiplex fluorescent PCR method) developed by Amoy Diagnostics in Xiamen. There is also the EGFR/ALK/BRAF/KRAS gene mutation detection equipment developed by Burning Rock in Guangzhou End-stop sequencing method) and the EGFR, KRAS, BRAF, PIK3CA, ALK, ROS1 gene mutation detection kit (semiconductor sequencing method) developed by Novogene in Tianjin which are used for multi-gene detection.
In vitro detection is mainly used for auxiliary diagnosis of tumors and companion diagnosis of anti-tumor drugs. For example, Septin9 gene methylation detection kit (PCR fluorescent probe method) provides a non-invasive auxiliary diagnosis method for colorectal cancer and the EGFR gene mutation detection kit (multiplex fluorescent PCR method) is the first companion diagnostic test product approved for osimertinib mesylate tablets in China. In addition, in vitro diagnostic products are also used for diagnosis of prenatal and postnatal care, diagnosis of rare diseases, and diagnosis of viruses and microorganisms. For example, the Survival Motor Neuron Gene 1 (SMN1) Exon Deletion Detection Kit (Fluorescence Quantitative PCR) is used for in vitro auxiliary molecular diagnosis of patients with spinal muscular atrophy (SMA).